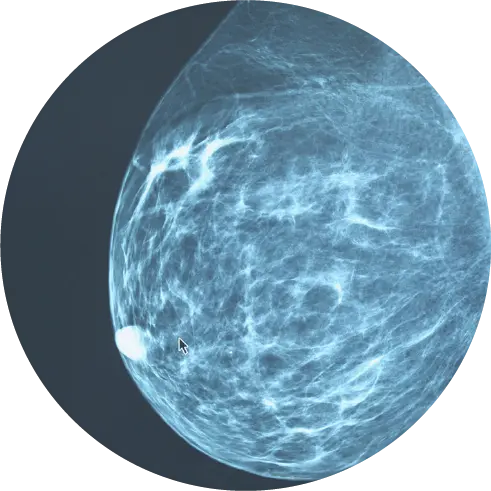
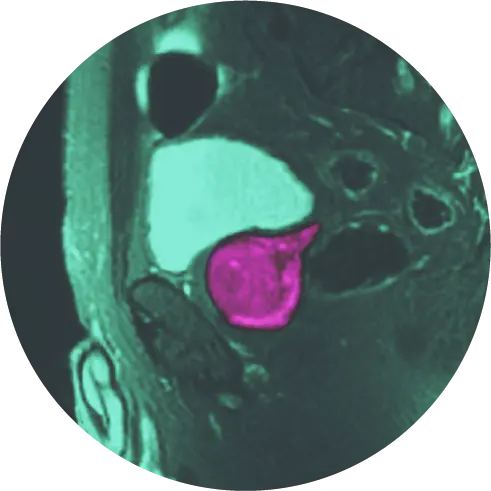
Almost 20 years in the making, researchers say PYLARIFY® is a triumph for the field of molecular and diagnostic imaging.
Another groundbreaking prostate cancer imaging tool is now available at HALO Diagnostics.
The tool is an injectable contrasting agent called PYLARIFY® that targets prostate-specific membrane antigen (PSMA), a protein found on the surface of 90 percent of primary and metastatic prostate cancer cells[I]. The agent, also known as piflufolastat F-18, or simply “PyL,” is FDA approved for use with targeted positron emission tomography (PET) imaging to identify suspected metastatic or recurrent prostate cancer.
What is Pylarify?
Developed by Lantheus, it is the only commercially available PSMA PET imaging agent for prostate cancer. Just a few months after receiving FDA approval, it is now part of the National Comprehensive Cancer Networks (NCCN) guidelines for prostate cancer imaging[II].
PYLARIFY is a radioactive tracer molecularly engineered to target PSMA like a heat-seeking missile. It bonds to the protein and causes it to light up on a PET scan, shining a virtual spotlight on even the tiniest bits of prostate cancer anywhere in the body.
What does it do?
Coupled with a HALO Dx PET scanner, PYLARIFY enables the reader to detect and locate metastasis in the lymph nodes, bone, and soft tissue.
The product is considered a breakthrough for men who were initially treated for localized prostate cancer who suspect the disease has returned. Of those men, up to 50 percent may experience disease recurrence within ten years[III]. Recurrent disease is often detected by rising PSA levels; however, conventional imaging, especially at low PSA levels, is not able to identify the location and extent of the disease in most cases[IV] [V].
“PYLARIFY is a molecular imaging game-changer for early detection of metastasis in newly diagnosed prostate cancer patients, and for recurrence in men previously treated for the disease,” said Dr. John Feller, HALO Diagnostics Chief Medical Officer. “Early and more precise detection saves lives and gives patients multiple treatment options.”
PYLARIFY can be used on both treatment-naive patients with advanced disease and recurrence patients even if there is no sign of metastasis on a bone scan or CT scan.
Clinical Trials
In two clinical trials, OSPREY and CONDOR, PYLARIFY was well tolerated. 593 patients with various states of prostate cancer were exposed to a single dose. Adverse reactions including headache and fatigue were reported in less than two percent of patients within the studies[VI][VII].
“Its an exciting time in the field,” said Dr. Feller. “These tools are invaluable when it comes to early detection and effective treatment planning. PSMA research is helping us understand so much more about metastatic prostate cancer.”
If you think you may benefit from a PSMA PET scan, schedule a consultation with a HALO Dx team member to discuss your diagnostic and/or treatment options. Call 760-469-8057, or click the button below.
Article references:
[I] Ceci & Fanti. PSMA-PET/CT imaging in prostate cancer: why and when. Clinical and Translational Imaging volume 7, pages377379 (2019).
[III] Alipour et al. Guiding management of therapy in prostate cancer: time to switch from conventional imaging to PSMA PET? Ther Adv Med Oncol. 2019; 11: 1758835919876828.
[IV] 1Mena et al. 18 F-DCFPyL PET/CT Imaging in Patients with Biochemically Recurrent Prostate Cancer After Primary Local Therapy J Nucl Med 2020 Jun;61(6):881-889. doi: 10.2967/jnumed.119.234799. Epub 2019 Nov 1.
[V] Alipour et al. Guiding management of therapy in prostate cancer: time to switch from conventional imaging to PSMA PET? Ther Adv Med Oncol. 2019; 11: 1758835919876828.
[VI] Pienta KJ, Gorin MA, Rowe SP, et al. A phase 2/3 prospective multicenter study of the diagnostic accuracy of prostate specific membrane antigen PET/CT with 18F-DCFPYL in prostate cancer patients (OSPREY) [published online ahead of print, February 26, 2021]. J Urol. doi:10.1097/JU.0000000000001698
[VII] Morris MJ, Rowe SP, Gorin MA, et al. Diagnostic performance of 18F-DCFPYL-PET/CT in men with biochemically recurrent prostate cancer: results from the CONDOR phase III, multicenter study [published online ahead of print, February 23, 2021]. Clin Cancer Res. doi:10.1158/1078-0432.CCR-20-4573
You should always consult with your doctor(s) when considering treatment options. HALO Dx provides MRI-guided prostate screenings, MRI-guided prostate biopsy, liquid “biopsy” tests, and genomic testing to facilitate informed decisions between doctors and patients. Learn more about HALO Dx’s advanced, minimally invasive prostate cancer and BPH treatments including Laser Focal Therapy and TULSA-PRO for whole gland and partial ablations. With the appropriate diagnostic results, our prostate disease experts help you answer any questions you may have about prostate cancer or BPH.